|
5/20/2023 0 Comments Ion bonding service
Stacee Kratovil joins Parcion as a Senior Family Advisor, bringing more than 10 years of experience working within the multi-family office and private wealth sectors to the firm. “I look forward to helping individuals and families create customized investment strategies that are right for them.” “I’m excited to be part of the Parcion team, and work with a group of professionals that share a commitment to serving clients in the best way possible,” said Bond. Outside of work, he enjoys spending time with his wife and four kids, biking, hiking, and golfing. He earned his bachelor’s degree from Purdue University and holds an MBA in Finance from Indiana University. Most recently, he was a private investment analyst and portfolio manager at Winthrop Capital Management. He also served as the chief investment officer at Gripman Investment Advisors, where he managed a multi-asset strategy focused on delivering superior risk adjusted returns for clients. Prior to joining Parcion, Bond worked at JP Morgan Asset Management for more than 10 years and managed more than $100 billion in assets across core, core plus, and insurance fixed income strategies. With extensive experience in building public and private security investment portfolios, Bond’s expertise will be integral in helping clients uncover key opportunities to accomplish their investment goals. In his role, Bond will handle day-to-day portfolio management, overseeing investment and trading strategies for the firm’s clients. “Tim and Stacee are tremendous additions, both bringing impressive levels of talent and experience to our firm, and a shared commitment to delivering world class service.” “One of our primary goals has been to build a team of passionate professionals with the common intent of delivering exceptional client results,” said Terry Cook, CFP®, CIMA®, Parcion CEO + Managing Principal. 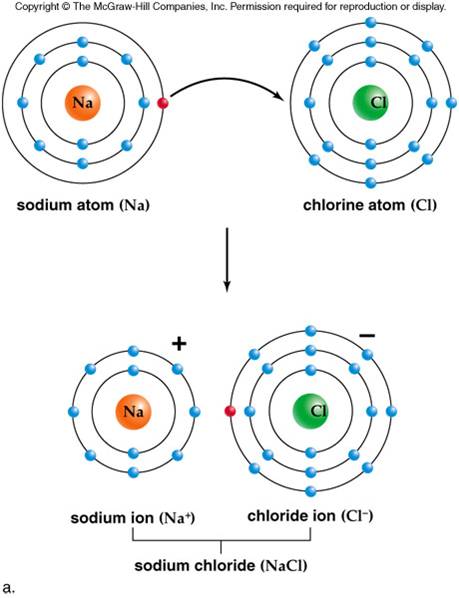
Ionic compounds cannot conduct electricity in the solid state because their ions are held in fixed positions and cannot move.Parcion Private Wealth, a Seattle-area wealth management firm focused on helping business owners and entrepreneurs optimize wealth events, recently welcomed Tim Bond as Director of Investment Operations and Portfolio Management and Stacee Kratovil as a Senior Family Advisor. it has dissolved in water to form an aqueous solutionīoth these processes allow ions to move from place to place.these particles are free to move from place to placeĪn ionic compound can conduct electricity when:.it contains charged particles, such as ions, and.Ionic bonds between Mg 2+ and O 2- ions are stronger than those between Na + and Cl - ions Conducting electricity Ions with higher charge will have stronger forces between them, so will need more energy in order to overcome these forces. The strength of the ionic bonds depends on the charge on the ions. 
As the ionic lattice contains such a large number of ions, a lot of energy is needed to overcome this ionic bonding so ionic compounds have high melting and boiling points. These forces are usually referred to as ionic bonding. Ionic compounds are held together by electrostatic forces between the oppositely charged ions. Since the electrostatic forces of attraction between oppositely charged ions are strong, their melting and boiling points are high. The more energy needed, the higher the melting point or boiling point. all remaining forces are overcome during boiling.some forces are overcome during melting. 
This energy overcomes the strong electrostatic forces of attraction which act in all directions between the oppositely charged ions: The three states of matter to see how bonding and structure are related to the properties of substances.Įnergy must be transferred to a substance to make it melt or boil. Ionic compounds have high melting and boiling points, so they are in the solid state at room temperature.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
